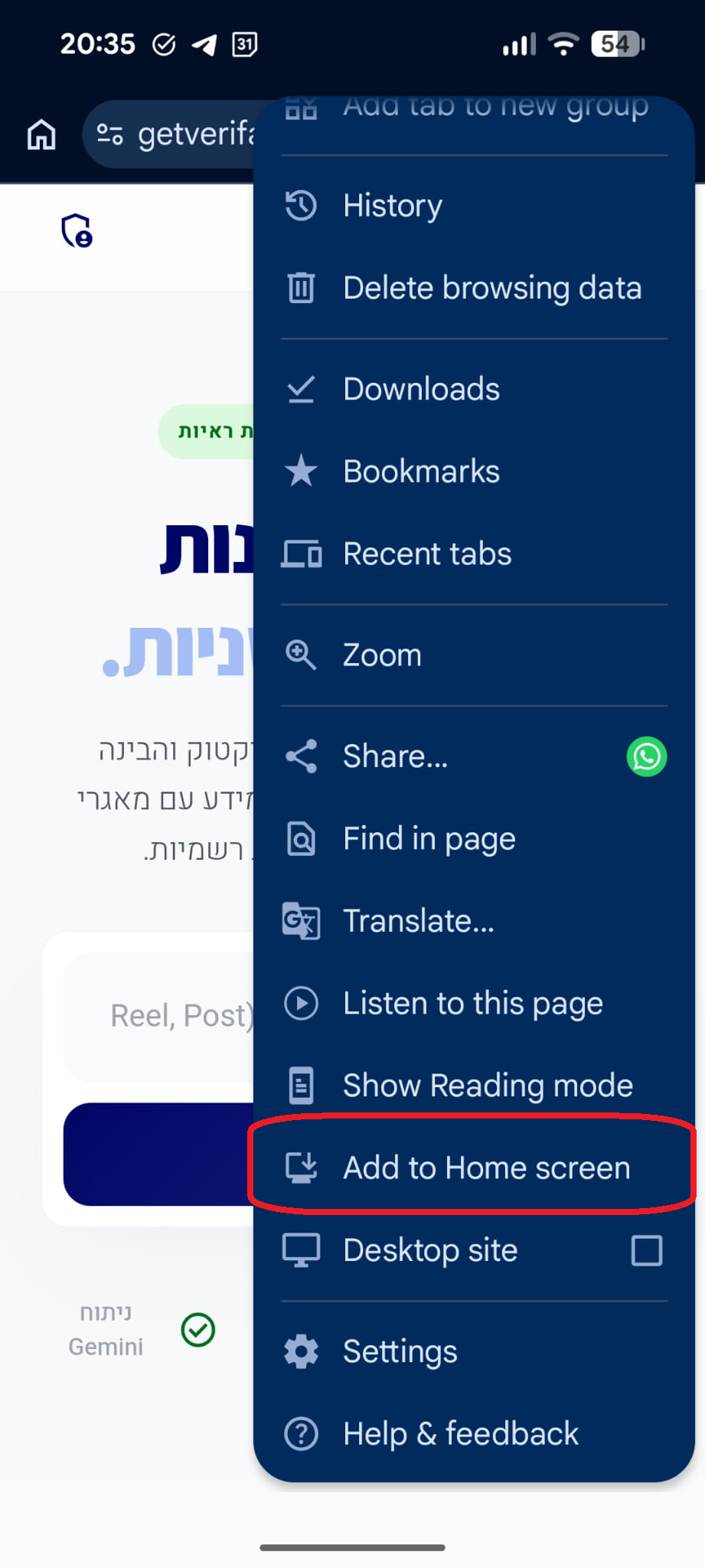
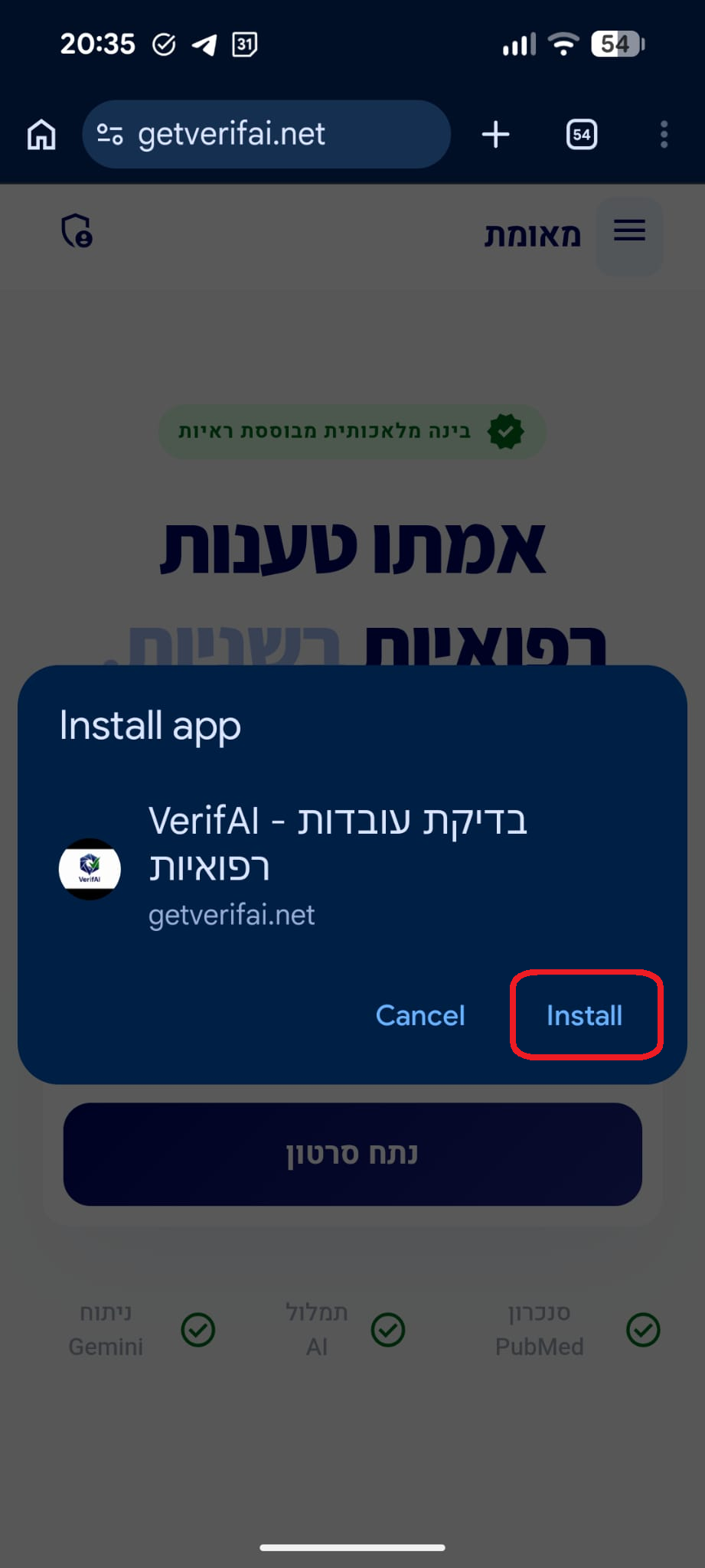
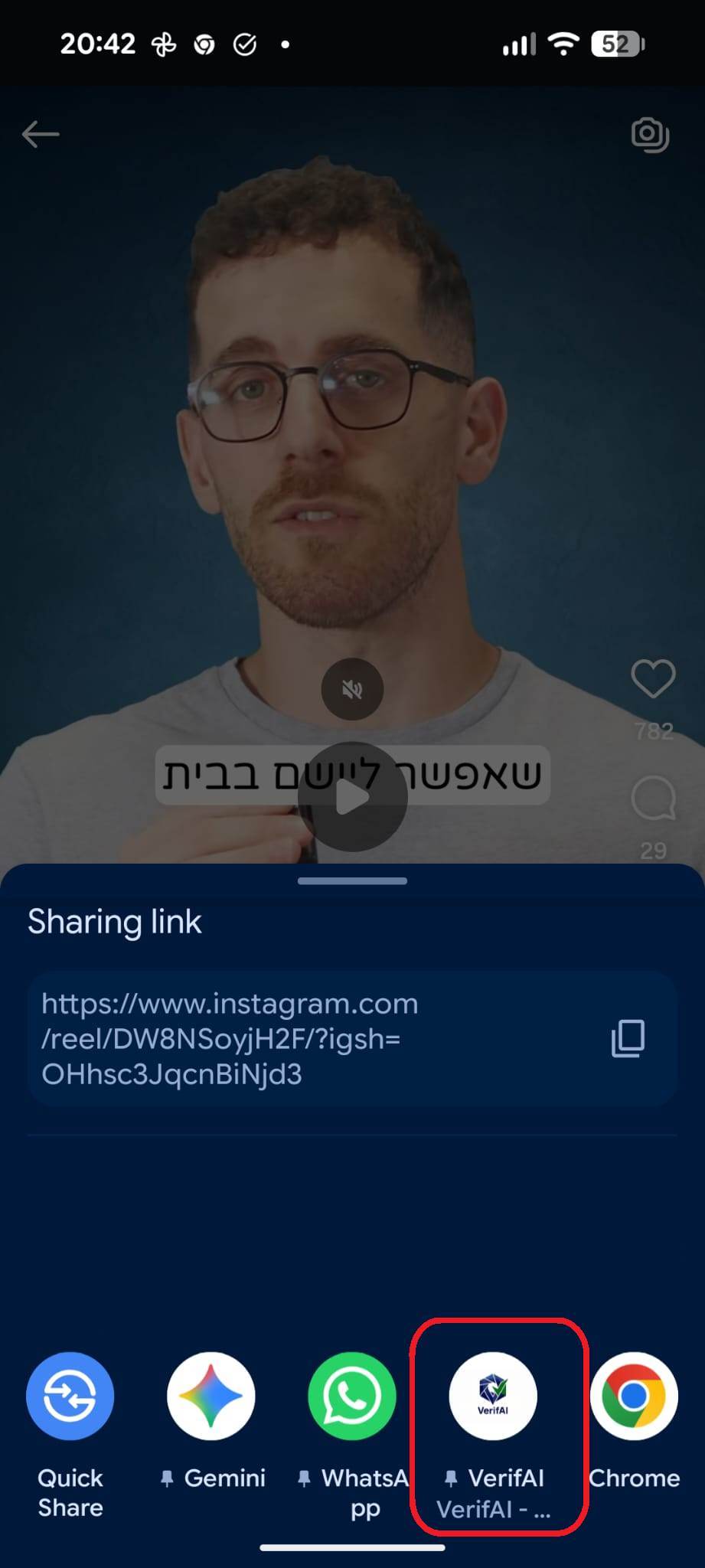
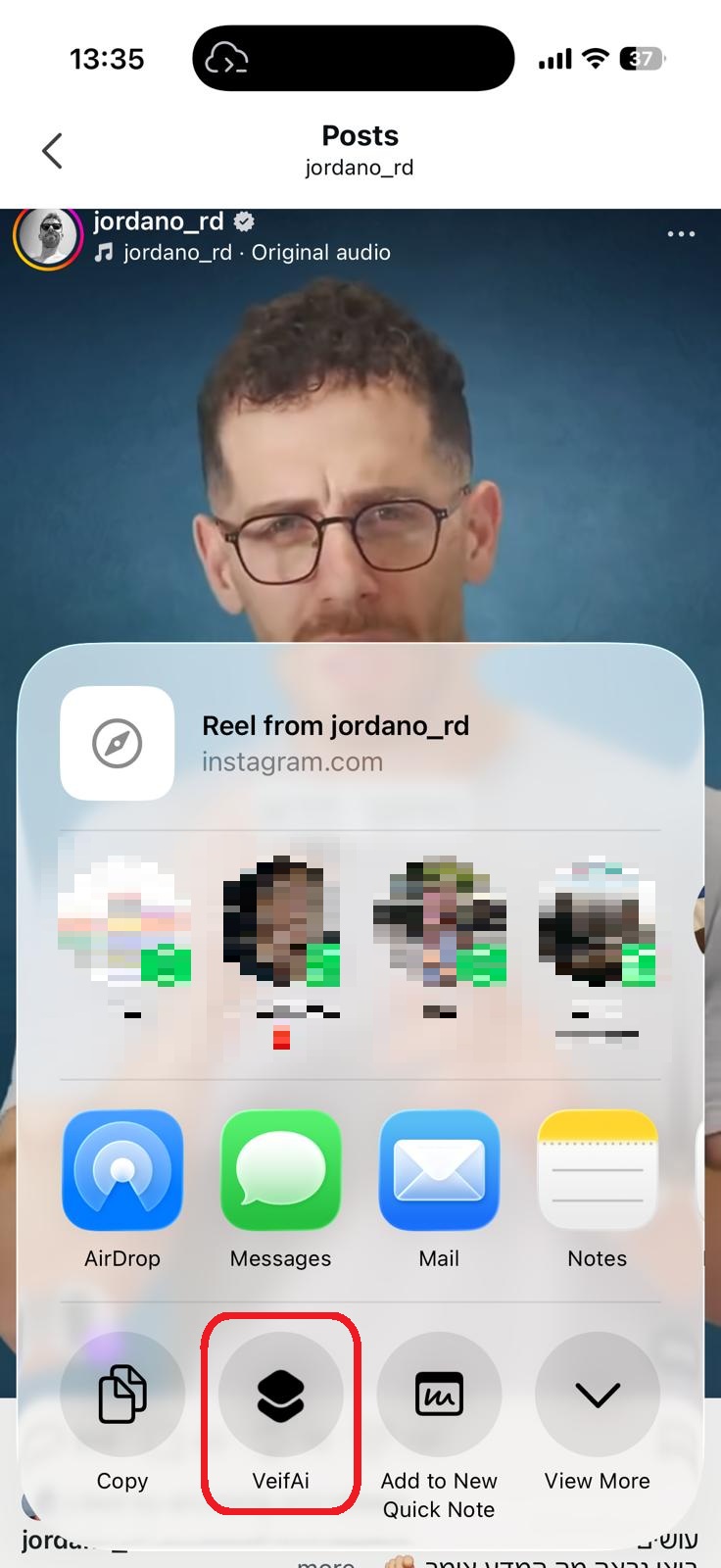
סריקה בתהליך...
הניתוח עשוי לארוך 30–90 שניות
הידעת?
הסרטון אינו מכיל תוכן רפואי
חושבים שחלה טעות?
אם לדעתכם הסרטון אכן עוסק בטענות רפואיות או בריאותיות, הוסיפו הסבר קצר ושלחו בקשה לבדיקה מחדש.
הבקשה התקבלה! נבדוק את הסרטון ונחזור אליכם.
אירעה שגיאה בשליחת הבקשה. נסו שוב.
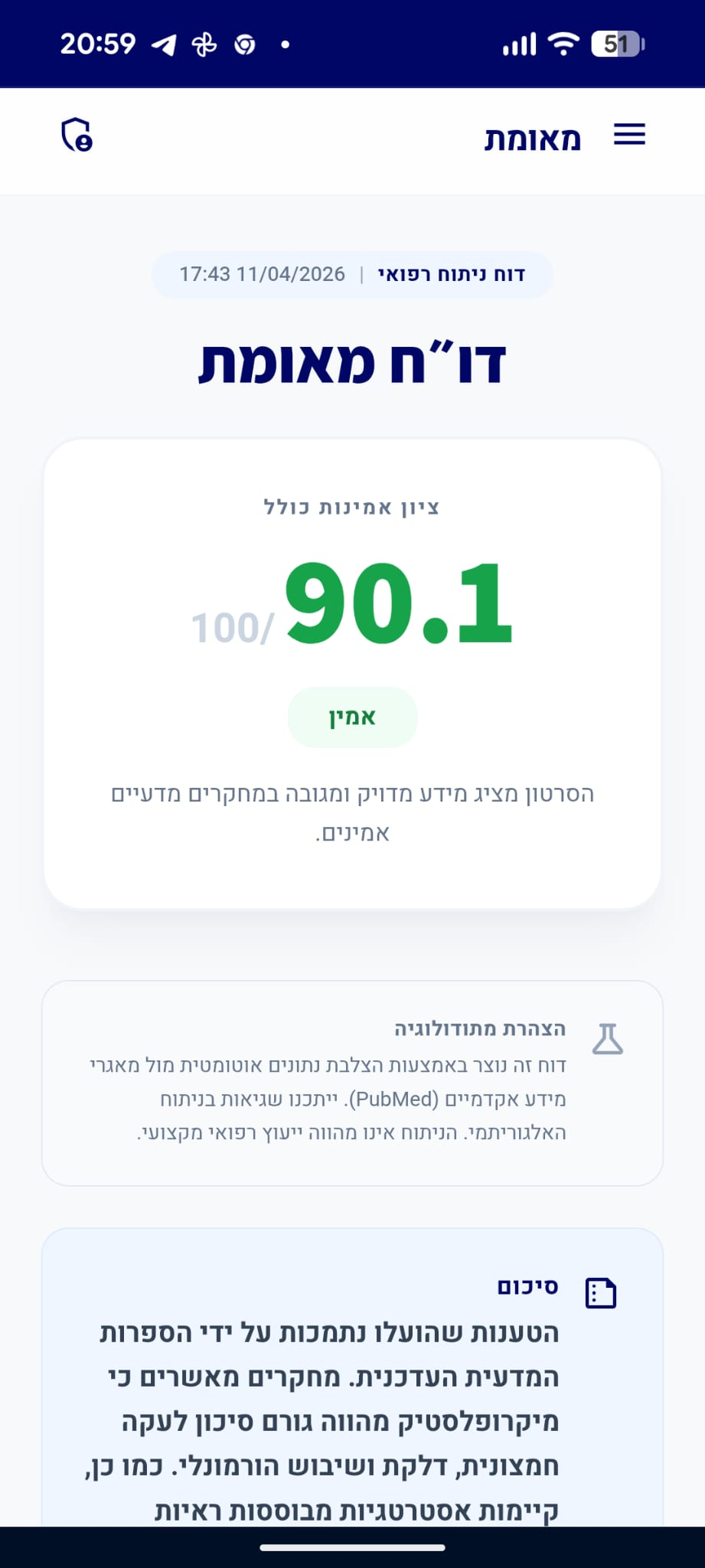
דו״ח מאומת
הסרטון מציג מידע מדויק ומגובה במחקרים מדעיים אמינים.
סיכום
הקליפ הציג טענה לפורמולה ייחודית לאיזון הורמונלי, בעוד המומחה הבהיר כי אין הוכחות מדעיות ליעילותן של פטריות בטיפול ב-PCOS וכי עדות אישית אינה תחליף למחקר קליני. המומחה צדק בהסבריו, שכן האבחנה של PCOS מבוססת על קריטריונים רפואיים קשיחים ואישור משרד הבריאות לתוספים אינו מעיד על יעילות רפואית. הראיות המדעיות תומכות בעמדת המומחה ומדגישות את הצורך בטיפול מבוסס ראיות ולא בהסתמכות על תוספים ללא תיקוף קליני.
תוכן פרסומי
מקדם: תוספי תזונה (פטריות מרפא) (בסרטון)
דוח על סרטון תגובה
סרטון זה מציג קליפ של אדם המביע טענות רפואיות, ומומחה/מגיב שמתייחס אליהן. הציון מבוסס רק על טענות המומחה.
quiz טענות הקליפ ותגובת המומחה
"התחלתי לקחת פטריות מרפא וקיבלתי מחזור אחרי חודשיים."
"אין מחקרים בבני אדם על הפטריות הללו בהקשר של תסמונת שחלות פוליציסטיות (PCOS)."
המומחה מפריך את הטענה שהפטריות הן טיפול מוכח ל-PCOS.
מסקנת הבדיקה:
נכון לעכשיו, אין מחקרים קליניים מבוססים בבני אדם המאשרים את יעילותן של פטריות ספציפיות כטיפול בתסמונת שחלות פוליציסטיות (PCOS). הספרות המדעית הקיימת מתמקדת במנגנונים ביולוגיים כלליים או בתוספים אחרים, אך לא בטיפול מבוסס פטריות למצב זה. (🟩)
"עדות אישית אינה הוכחה ליעילות טיפול, שכן מחזורים עשויים להופיע ב-PCOS גם ללא התערבות."
המומחה מסביר מדוע הופעת המחזור לאחר נטילת התוסף אינה מעידה על קשר סיבתי.
מסקנת הבדיקה:
עדות אישית אינה מהווה הוכחה מדעית ליעילות טיפול, שכן ב-PCOS עשויים להופיע מחזורים באופן ספונטני או כתוצאה משינויים באורח חיים, ללא קשר לתוסף ספציפי שנלקח. (🟩)
"יש לי פורמולה ייחודית לאיזון הורמונלי."
info המומחה לא התייחס ישירות לטענה זו ואין מספיק נתונים לבדיקה עצמאית
person_check טענות נוספות של המומחה
טענות שהמומחה הביע שאינן קשורות ישירות לטענה ספציפית בקליפ
"אישור משרד הבריאות לתוסף מעיד על תקינות המפעל בלבד, ולא על יעילות או בטיחות קלינית של המוצר."
מסקנת הבדיקה:
אישור משרד הבריאות לתוספי תזונה מתמקד בדרך כלל בבטיחות הייצור, תנאי המפעל ועמידה בתקני תברואה, ולא מהווה אישור קליני ליעילות רפואית או בטיחות ארוכת טווח של המוצר כפי שנדרש מתרופות. (🟩)
chevron_right מקורות מדעיים: (2)
-
link
Regulation of Food Supplements and Pharmacists' Responsibility in Professional Practice: A Review.
(1) Background: Regulations governing food supplements vary considerably across countries, allowing products that are prohibited in one jurisdiction to be legally sold in another. Furthermore, online sales enable and facilitate this practice. Regarding pharmaceutical malpractice, the absence of a standardized European framework complicates the evaluation of pharmacist liability. As a result, the specific elements of the liability framework are defined by the national legislation of each Member State. The aim of our review is to map the global regulatory landscape of food supplements and to examine the pharmacist's professional responsibilities, including instances of malpractice related to this area. (2) Methods: A literature review covering publications from January 2020 to December 2024 was performed using four databases: Scopus, PubMed, Embase, and Web of Science. The search retrieved 8243 records, of which 77 studies fulfilled the eligibility criteria. The extracted data were organized into five main themes: pharmacist responsibility and malpractice, food supplement regulation, consumer safety, health claims, and pharmacist knowledge. (3) Results: The literature reviewed indicated a relatively low number of malpractice cases within the pharmacy profession compared to other professions. A higher incidence of cases is observed among male pharmacists and those practicing in the private sector. Notably, no cases have been identified addressing pharmacists' responsibilities in the dispensing of food supplements. In the context of food supplement regulation, the reviewed literature highlights a lack of standardized terminology and harmonized legislation across different jurisdictions. Therefore, products may be classified differently across jurisdictions. Another observed barrier is the considerable variation in market access requirements across countries. Regarding consumer safety, several irregularities have been observed. Substantial non-compliance in both product composition and labeling has been observed, reflecting insufficient quality control measures. Concerning health claims, significant regulatory non-compliance with European Union regulations has been documented. In addition, widespread misleading advertising practices have been observed. With respect to pharmacists' knowledge, the reviewed literature identifies several professional challenges within pharmacy practice, particularly those concerning the dispensing of food supplements. (4) Conclusions: This research offers a comprehensive analysis of the literature published over the past five years concerning pharmaceutical malpractice cases, as well as an examination of food supplement regulation and the professional responsibilities of pharmacists. A recurring barrier identified is the absence of unified regulatory frameworks worldwide. This results in uncertainty concerning the pharmacist's professional role and responsibilities.…
PMID: 41718226
-
link
Scientific Validation of "Clinically Proven" Claims: Compliance Focused and Global Regulatory Insights.
The designation "clinically proven" represents a product's validation through scientifically rigorous, ethically sound, and regulatory-compliant clinical research. Establishing such a claim demands a systematic continuum from preclinical evaluations to well-designed randomized controlled trials (RCTs), multicentric validations, and real-world evidence studies that collectively demonstrate safety, efficacy, and reproducibility. This communication delineates the essential methodological and regulatory framework required to substantiate clinically proven claims. It underscores the critical role of robust RCTs, investigator-initiated trials, post-marketing surveillance, and global real-world studies encompassing diverse populations, standardized endpoints, and strict adherence to Good Clinical Practice (GCP) guidelines. Furthermore, it highlights the influence of geographical variability, investigator expertise, and environmental factors on study outcomes. The regulatory perspectives of key authorities, including the Central Drugs Standard Control Organization (CDSCO), the United States Food and Drug Administration (USFDA), the Therapeutic Goods Administration (TGA), and the European Medicines Agency (EMA), are discussed in relation to maintaining the authenticity and credibility of such claims. Emerging technologies such as artificial intelligence-assisted imaging, advanced instrumental analyses, and digital data monitoring are identified as pivotal tools enhancing evidence reliability. Ultimately, the concept of "clinically proven" extends beyond empirical validation, embodying scientific integrity, ethical transparency, and consumer trust at the intersection of innovation and regulatory compliance.…
PMID: 41625816
"תסמונת שחלות פוליציסטיות היא אבחנה רפואית המבוססת על קריטריונים ספציפיים (מחזורים לא סדירים, עודף אנדרוגנים, או מראה אופייני באולטרסאונד)."
מסקנת הבדיקה:
האבחנה של תסמונת שחלות פוליציסטיות (PCOS) מבוססת אכן על קריטריונים רפואיים מוגדרים (קריטריוני רוטרדם), הכוללים הפרעות בביוץ, עודף אנדרוגנים קליני או ביוכימי, ומראה שחלות פוליציסטיות באולטרסאונד. (🟩)
chevron_right מקורות מדעיים: (3)
-
link
AMERICAN ASSOCIATION OF CLINICAL ENDOCRINOLOGISTS, AMERICAN COLLEGE OF ENDOCRINOLOGY, AND ANDROGEN EXCESS AND PCOS SOCIETY DISEASE STATE CLINICAL REVIEW: GUIDE TO THE BEST PRACTICES IN THE EVALUATION AND TREATMENT OF POLYCYSTIC OVARY SYNDROME--PART 1.
Polycystic Ovary Syndrome (PCOS) is recognized as the most common endocrine disorder of reproductive-aged women around the world. This document, produced by the collaboration of the American Association of Clinical Endocrinologists (AACE) and the Androgen Excess and PCOS Society (AES) aims to highlight the most important clinical issues confronting physicians and their patients with PCOS. It is a summary of current best practices in 2015. PCOS has been defined using various criteria, including menstrual irregularity, hyperandrogenism, and polycystic ovary morphology (PCOM). General agreement exists among specialty society guidelines that the diagnosis of PCOS must be based on the presence of at least two of the following three criteria: chronic anovulation, hyperandrogenism (clinical or biological) and polycystic ovaries. There is need for careful clinical assessment of women's history, physical examination, and laboratory evaluation, emphasizing the accuracy and validity of the methodology used for both biochemical measurements and ovarian imaging. Free testosterone (T) levels are more sensitive than the measurement of total T for establishing the existence of androgen excess and should be ideally determined through equilibrium dialysis techniques. Value of measuring levels of androgens other than T in patients with PCOS is relatively low. New ultrasound machines allow diagnosis of PCOM in patients having at least 25 small follicles (2 to 9 mm) in the whole ovary. Ovarian size at 10 mL remains the threshold between normal and increased ovary size. Serum 17-hydroxyprogesterone and anti-Müllerian hormone are useful for determining a diagnosis of PCOS. Correct diagnosis of PCOS impacts on the likelihood of associated metabolic and cardiovascular risks and leads to appropriate intervention, depending upon the woman's age, reproductive status, and her own concerns. The management of women with PCOS should include reproductive function, as well as the care of hirsutism, alopecia, and acne. Cycle length >35 days suggests chronic anovulation, but cycle length slightly longer than normal (32 to 35 days) or slightly irregular (32 to 35-36 days) needs assessment for ovulatory dysfunction. Ovulatory dysfunction is associated with increased prevalence of endometrial hyperplasia and endometrial cancer, in addition to infertility. In PCOS, hirsutism develops gradually and intensifies with weight gain. In the neoplastic virilizing states, hirsutism is of rapid onset, usually associated with clitoromegaly and oligomenorrhea. Girls with severe acne or acne resistant to oral and topical agents, including isotretinoin (Accutane), may have a 40% likelihood of developing PCOS. Hair loss patterns are variable in women with hyperandrogenemia, typically the vertex, crown or diffuse pattern, whereas women with more severe hyperandrogenemia may see bitemporal hair loss and loss of the frontal hairline. Oral contraceptives (OCPs) can effectively lower androgens and block the effect of androgens via suppression of ovarian androgen production and by increasing sex hormone-binding globulin. Physiologic doses of dexamethasone or prednisone can directly lower adrenal androgen output. Anti-androgens can be used to block the effects of androgen in the pilosebaceous unit or in the hair follicle. Anti-androgen therapy works through competitive antagonism of the androgen receptor (spironolactone, cyproterone acetate, flutamide) or inhibition of 5α-reductase (finasteride) to prevent the conversion of T to its more potent form, 5α-dihydrotestosterone. The choice of antiandrogen therapy is guided by symptoms. The diagnosis of PCOS in adolescents is particularly challenging given significant age and developmental issues in this group. Management of infertility in women with PCOS requires an understanding of the pathophysiology of anovulation as well as currently available treatments. Many features of PCOS, including acne, menstrual irregularities, and hyperinsulinemia, are common in normal puberty. Menstrual irregularities with anovulatory cycles and varied cycle length are common due to the immaturity of the hypothalamic-pituitary-ovarian axis in the 2- to 3-year time period post-menarche. Persistent oligomenorrhea 2 to 3 years beyond menarche predicts ongoing menstrual irregularities and greater likelihood of underlying ovarian or adrenal dysfunction. In adolescent girls, large, multicystic ovaries are a common finding, so ultrasound is not a first-line investigation in women <17 years of age. Ovarian dysfunction in adolescents should be based on oligomenorrhea and/or biochemical evidence of oligo/anovulation, but there are major limitations to the sensitivity of T assays in ranges applicable to young girls. Metformin is commonly used in young girls and adolescents with PCOS as first-line monotherapy or in combination with OCPs and anti-androgen medications. In lean adolescent girls, a dose as low as 850 mg daily may be effective at reducing PCOS symptoms; in overweight and obese adolescents, dose escalation to 1.5 to 2.5 g daily is likely required. Anti-androgen therapy in adolescents could affect bone mass, although available short-term data suggest no effect on bone loss.…
PMID: 26509855
-
link
PCOS in Adolescents-Ongoing Riddles in Diagnosis and Treatment.
Polycystic ovary syndrome (PCOS) is one of the most common endocrine disorders in women of reproductive age. A diagnosis of PCOS is established when a patient exhibits two of three Rotterdam criteria: oligoovulation or anovulation, excess androgen activity, and polycystic ovarian morphology. The pathogenesis of PCOS, as it affects adolescents, is often discussed in terms of a "two-hit" theory. This refers to a stepwise process in which the first "hit" is an inborn congenitally programmed predisposition, while the second "hit" arises from a provocative factor such as insulin resistance. The dynamic physiological and anatomical changes which occur in puberty make for a challenging diagnosis in this group of patients. It is important to be mindful of the physiological particularities in adolescence which often mimic the symptoms of PCOS. In their first-year post-menarche, approximately 75% of menstruating adolescents report their cycle to last between 21-45 days. Recent studies have shown that regular menstrual cyclicity is only achieved within 2-3 years post-menarche. Anovulation, as a crucial diagnostic element for PCOS, features in about half of early-post-menarchal adolescents. Hirsutism and acne are the most common clinical manifestations of hyperandrogenism, and mild features are developed by most adolescents as a result of elevated androgen levels. Distinguishing between a pathological sign and normal features of maturation is often difficult. A polycystic ovarian morphology (PCOM) through ultrasound has been found in up to 40%, 35%, and 33.3% of patients when assessed at 2, 3, and 4 years, respectively, after menarche. PCOM in adolescence is not associated with future abnormalities in ovulatory rate or menstrual cycle duration. For this reason, international guidelines recommend against the use of pelvic ultrasound until 8 years post-menarche. The primary aim of management is focused mainly on improving hormonal and metabolic status, the prevention of future comorbid complications, and generally improving the overall quality of life in young women with PCOS. Considerable controversy surrounds the choice of optimal pharmacological treatment to address PCOS in adolescents. Reliable studies, which include this sub-section of the population, are very limited. There is a lack of robust and reliable trials in the literature addressing the use of combined oral contraceptives. Further work needs to be undertaken in order to provide safe and effective care to the adolescent population in this regard.…
PMID: 36769869
-
link
Diagnosis of Polycystic Ovary Syndrome With Predictive Modeling of Select Clinical Features.
<h4>Objective</h4>To determine whether a limited set of ultrasonographic, biochemical, and clinical features are sufficient to accurately predict polycystic ovary syndrome (PCOS) diagnosis.<h4>Methods</h4>Transvaginal ultrasound images and available clinical data for participants with PCOS (n=101) and controls (n=50) were used for this multicenter, retrospective pilot study. Diagnosis of PCOS was defined by the 2023 International Evidence-Based Guideline. Controls had no diagnostic features of PCOS. Differences in demographic (age, body mass index [BMI]), ultrasonographic (ovarian volume, follicle number per ovary, follicle number per single cross section), biochemical (sex hormone binding globulin, total testosterone, free androgen index, bioavailable testosterone), and clinical (follicle-stimulating hormone, luteinizing hormone, estradiol, anti-müllerian hormone (AMH), age at menarche, minimum self-reported menstrual cycle length in the past year, maximum self-reported menstrual cycle length in the past year, Ferriman-Gallwey hirsutism score) features between groups were assessed with Mann-Whitney <i>U</i> tests. A logistic regression model was trained to predict PCOS diagnosis using subsets of ultrasonographic, biochemical, and clinical variables. Model performance was evaluated with area under the receiver operating characteristic curve (AUROC) and F1 score measures.<h4>Results</h4>Anti-müllerian hormone alone predicted PCOS diagnosis with relatively good diagnostic accuracy (AUROC 0.884, F1 score 0.807). Integration of AMH and ovarian volume improved model performance (AUROC 0.906, F1 score 0.811). Integration of all ultrasonographic, biochemical, and clinical features yielded a high-performing model with excellent diagnostic accuracy for PCOS (AUROC 0.991, F1 score 0.811). Refinement of the model to a limited set of readily obtained variables, including AMH, ovarian volume, hirsutism score, and maximum self-reported menstrual cycle length, yielded a model with strong performance (AUROC 0.982, F1 score 0.805).<h4>Conclusion</h4>A minimum combination of ovarian volume with AMH and examination/history-derived end points can accurately predict PCOS diagnosis with performance comparable to that of a combination of all ultrasonographic, biochemical, and clinical features. This may streamline diagnostic workflows, thereby reducing clinical burden.<h4>Parent protocol clinical trial registration</h4>ClinicalTrials.gov, NCT01859663, NCT01927471, NCT03306849, NCT01927432, NCT03547453.…
PMID: 41868592

Inna Kreinin Bleicher
דירוג זה מבוסס על 2 דוחות אימות קודמים.
האם הדוח הזה היה מועיל לך?
מה היה פחות טוב? (רשות)
תודה על הפידבק!
עירעור על דוח זה
ספקו ראיות חדשות או הצביעו על אי דיוקים
נעדכן אותך על תוצאות הבדיקה
הוסיפו קישורים למחקרים או מקורות רפואיים מוכרים
העירעור נשלח בהצלחה!
המנוע המדעי שלנו יבדוק את הראיות שהגשתם. נעדכן אתכם באימייל עם התוצאות.
ניתוח מבוסס בינה מלאכותית
דוח זה נוצר באופן אוטומטי על ידי מערכת בינה מלאכותית ועשוי להכיל שגיאות, אי-דיוקים או מידע חלקי. הניתוח אינו מהווה ייעוץ רפואי, אבחנה או המלצה לטיפול, והוא אינו תחליף לדעתו של איש מקצוע רפואי מוסמך. יש להתייעץ עם רופא או מומחה מוסמך לפני קבלת כל החלטה רפואית. המידע מוצג לצרכי מידע כללי בלבד.
מידע זה מופק על ידי בינה מלאכותית ואינו מהווה תחליף לייעוץ רפואי מקצועי.