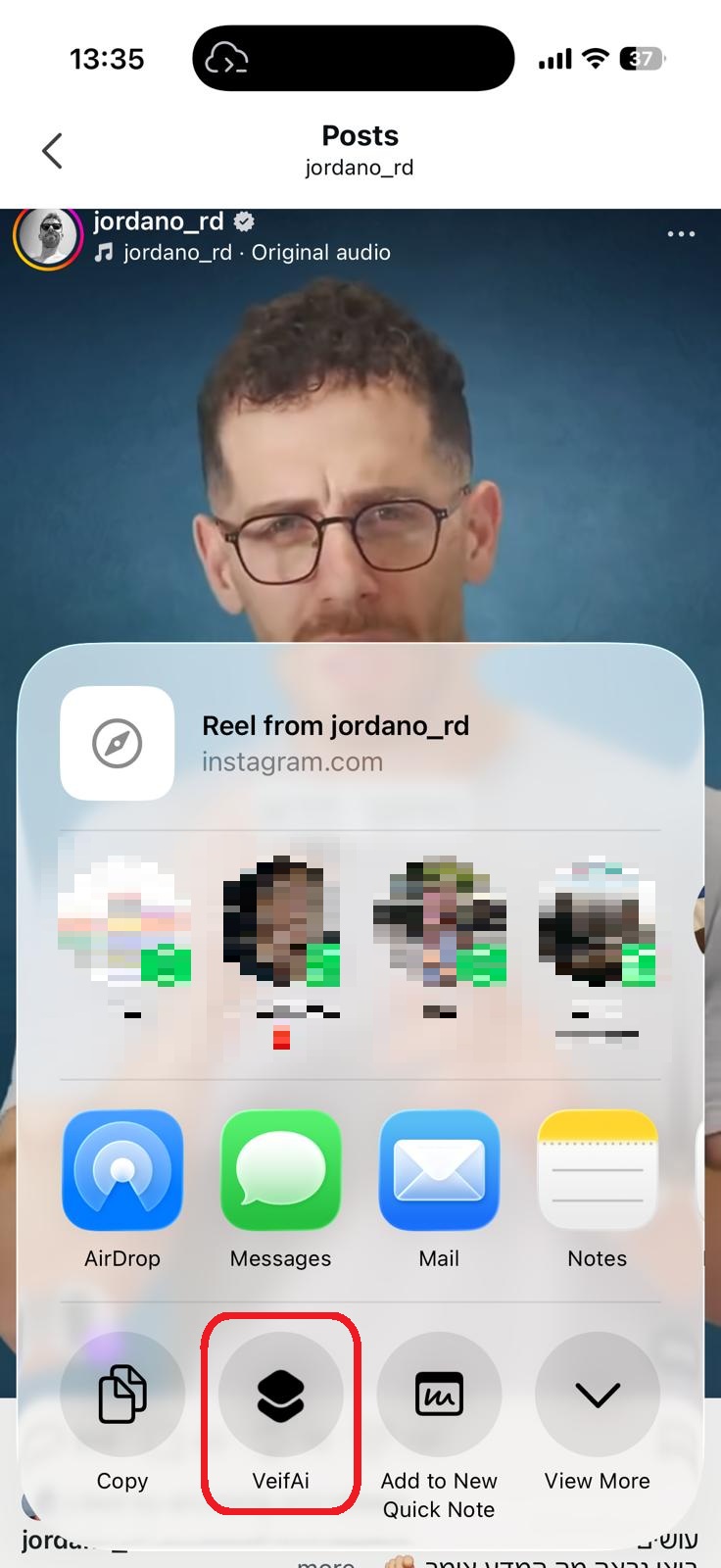
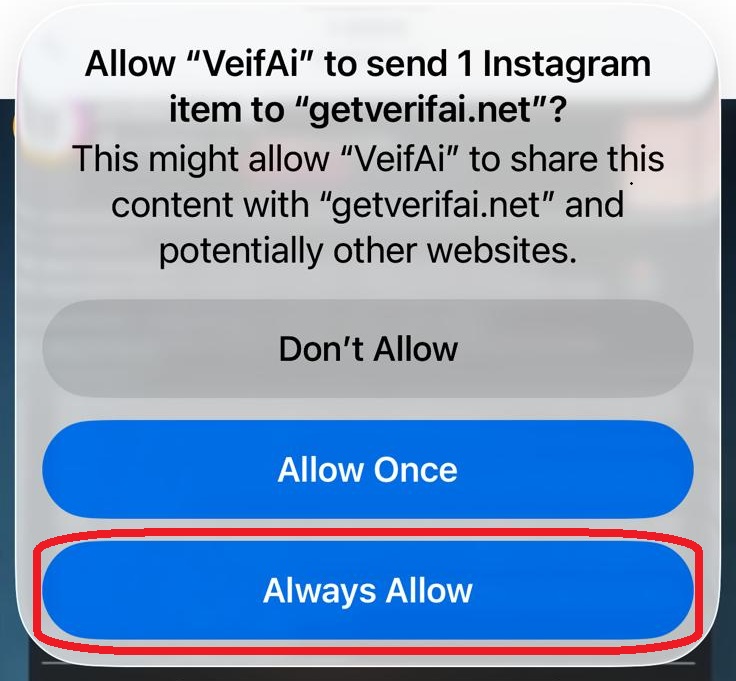
סריקה בתהליך...
הניתוח עשוי לארוך 30–90 שניות
הידעת?
הסרטון אינו מכיל תוכן רפואי
חושבים שחלה טעות?
אם לדעתכם הסרטון אכן עוסק בטענות רפואיות או בריאותיות, הוסיפו הסבר קצר ושלחו בקשה לבדיקה מחדש.
הבקשה התקבלה! נבדוק את הסרטון ונחזור אליכם.
אירעה שגיאה בשליחת הבקשה. נסו שוב.
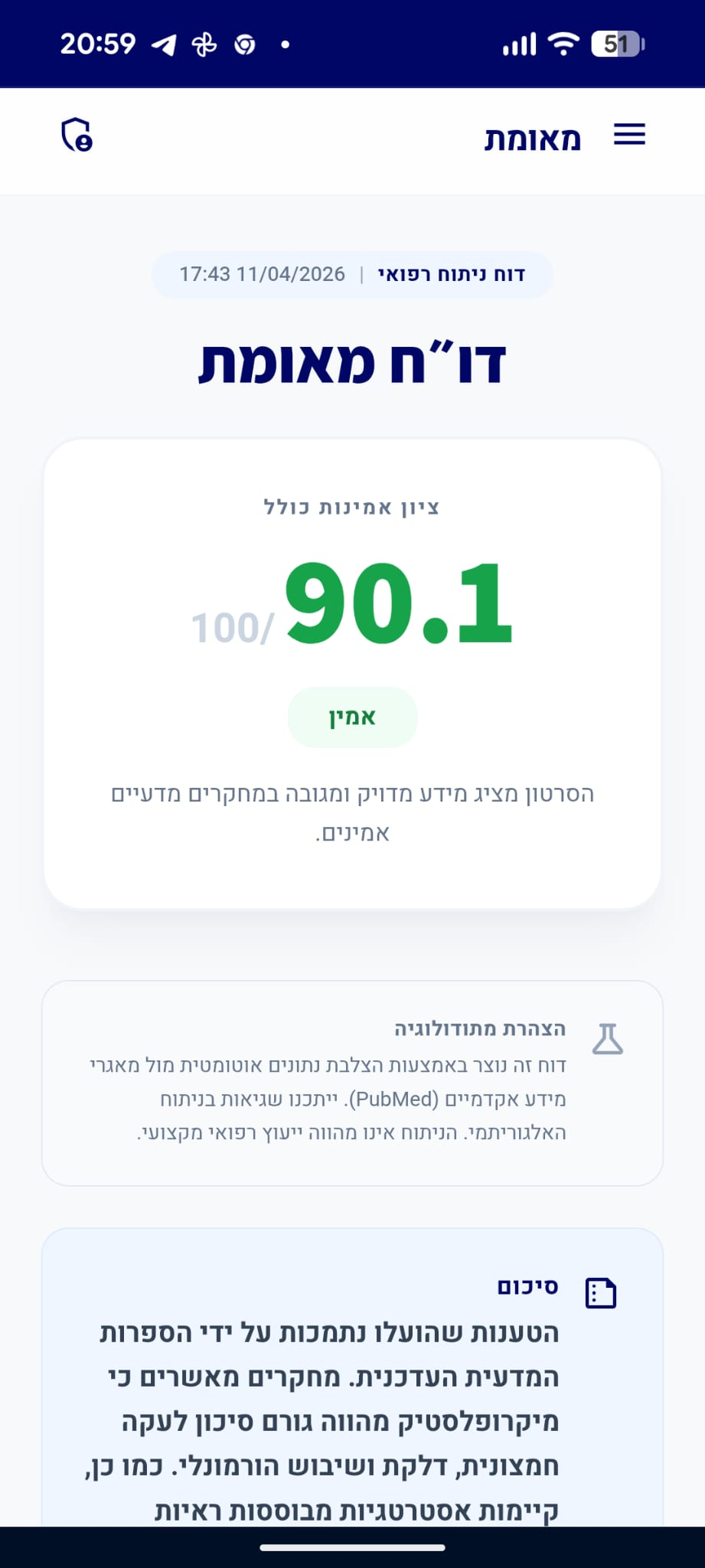
דו״ח מאומת
הסרטון מציג מידע מדויק ומגובה במחקרים מדעיים אמינים.
סיכום
הטענות שהועלו לגבי הרכבו הכימי של האספרטיים, תהליך פירוקו בגוף והנחיות ה-FDA לגבי צריכתו המותרת מאומתות על ידי הספרות המדעית. כמו כן, המקור ההיסטורי לחששות לגבי קשר לסרטן אכן מיוחס למחקרים מוקדמים בבעלי חיים. עם זאת, הטענה כי אין כל עדות לקשר בין אספרטיים לסרטן בבני אדם נחשבת למוטלת בספק, שכן מחקרים תצפיתיים מודרניים העלו ממצאים המצריכים בחינה נוספת, גם אם לא הוכחה סיבתיות חד-משמעית.
analytics ניתוח טענות מבוסס ראיות
"מחקרים שנערכו בבני אדם הראו כי אין הוכחה לכך שאספרטיים גורם לסרטן."
מסקנת הבדיקה:
בעוד שרוב המחקרים האפידמיולוגיים הגדולים לא מצאו קשר סיבתי מובהק בין צריכת אספרטיים לסרטן בבני אדם, קיימים מחקרים תצפיתיים עדכניים (כגון NutriNet-Santé) המצביעים על קשרים אפשריים, מה שהופך את הטענה ש'אין כל עדות' למורכבת ושנויה במחלוקת בקהילה המדעית. (🟨)
chevron_right מקורות מדעיים: (3)
-
link
Non-sugar sweeteners and cancer: Toxicological and epidemiological evidence.
Several toxicological and epidemiological studies were published during the last five decades on non-sugar sweeteners (NSS) and cancer. Despite the large amount of research, the issue still continues to be of interest. In this review, we provided a comprehensive quantitative review of the toxicological and epidemiological evidence on the possible relation between NSS and cancer. The toxicological section includes the evaluation of genotoxicity and carcinogenicity data for acesulfame K, advantame, aspartame, cyclamates, saccharin, steviol glycosides and sucralose. The epidemiological section includes the results of a systematic search of cohort and case-control studies. The majority of the 22 cohort studies and 46 case-control studies showed no associations. Some risks for bladder, pancreas and hematopoietic cancers found in a few studies were not confirmed in other studies. Based on the review of both the experimental data on genotoxicity or carcinogenicity of the specific NSS evaluated, and the epidemiological studies it can be concluded that there is no evidence of cancer risk associated to NSS consumption.…
PMID: 36870410
-
link
Can Artificial Sweeteners Increase the Risk of Cancer Incidence and Mortality: Evidence from Prospective Studies.
Cancer has become a major challenge in the global disease burden. Artificial sweeteners are a class of chemical compounds that are used as food and beverage addition agent to replace sugar. However, the health effects of consuming artificial sweeteners are still unclear. This meta-analysis was performed to evaluate the role of artificial sweeteners on cancer. The databases PubMed, Cochrane Library, MEDLINE, Web of Science and EMBASE were searched up until July 2022. A Newcastle−Ottawa scale (NOS) was used to estimate the study quality. A total of 25 observational studies were included with a total of 3,739,775 subjects. The intake of artificial sweeteners had no apparent association with overall cancer incidence and mortality. However, in Europe, artificial sweeteners’ intake could increase the risk of cancer incidence (HR/RR = 1.07, 95% CI = [1.02, 1.12], I2 = 25.8%, P = 0.223), which appears to be related to a shift in nutritional behaviors in the countries. Significant results were also observed in subgroups with aspartame and a mixed intake of artificial sweeteners. Moreover, higher risk was observed for artificial sweeteners intake in all-cause mortality (HR/RR =1.13, 95% CI = [1.03, 1.25], I2 = 79.7%, p < 0.001) and a J-shaped association between them was found. More data from well-conducted studies and clinical trials are required.…
PMID: 36145117
-
link
Artificial sweeteners and cancer risk: Results from the NutriNet-Santé population-based cohort study.
The food industry uses artificial sweeteners in a wide range of foods and beverages as alternatives to added sugars, for which deleterious effects on several chronic diseases are now well established. The safety of these food additives is debated, with conflicting findings regarding their role in the aetiology of various diseases. In particular, their carcinogenicity has been suggested by several experimental studies, but robust epidemiological evidence is lacking. Thus, our objective was to investigate the associations between artificial sweetener intakes (total from all dietary sources, and most frequently consumed ones: aspartame [E951], acesulfame-K [E950], and sucralose [E955]) and cancer risk (overall and by site). Overall, 102,865 adults from the French population-based cohort NutriNet-Santé (2009-2021) were included (median follow-up time = 7.8 years). Dietary intakes and consumption of sweeteners were obtained by repeated 24-hour dietary records including brand names of industrial products. Associations between sweeteners and cancer incidence were assessed by Cox proportional hazards models, adjusted for age, sex, education, physical activity, smoking, body mass index, height, weight gain during follow-up, diabetes, family history of cancer, number of 24-hour dietary records, and baseline intakes of energy, alcohol, sodium, saturated fatty acids, fibre, sugar, fruit and vegetables, whole-grain foods, and dairy products. Compared to non-consumers, higher consumers of total artificial sweeteners (i.e., above the median exposure in consumers) had higher risk of overall cancer (n = 3,358 cases, hazard ratio [HR] = 1.13 [95% CI 1.03 to 1.25], P-trend = 0.002). In particular, aspartame (HR = 1.15 [95% CI 1.03 to 1.28], P = 0.002) and acesulfame-K (HR = 1.13 [95% CI 1.01 to 1.26], P = 0.007) were associated with increased cancer risk. Higher risks were also observed for breast cancer (n = 979 cases, HR = 1.22 [95% CI 1.01 to 1.48], P = 0.036, for aspartame) and obesity-related cancers (n = 2,023 cases, HR = 1.13 [95% CI 1.00 to 1.28], P = 0.036, for total artificial sweeteners, and HR = 1.15 [95% CI 1.01 to 1.32], P = 0.026, for aspartame). Limitations of this study include potential selection bias, residual confounding, and reverse causality, though sensitivity analyses were performed to address these concerns. In this large cohort study, artificial sweeteners (especially aspartame and acesulfame-K), which are used in many food and beverage brands worldwide, were associated with increased cancer risk. These findings provide important and novel insights for the ongoing re-evaluation of food additive sweeteners by the European Food Safety Authority and other health agencies globally. ClinicalTrials.gov NCT03335644.…
PMID: 35324894
"אספרטיים הוא די-פפטיד המורכב משתי חומצות אמינו: חומצה אספרטית ופנילאלנין."
מסקנת הבדיקה:
אספרטיים הוא אכן די-פפטיד המורכב מחומצות האמינו חומצה אספרטית ופנילאלנין, כפי שמאושר בספרות המדעית ובתיאור המבנה הכימי שלו. (🟩)
chevron_right מקורות מדעיים: (2)
-
link
Aspartame--a sweet surprise.
The dipeptide ester L-aspartyl-L-phenylalanine methyl ester (APM) has been found to have a remarkably clean, sucrose-like taste with no off flavor and a potency 150-200 times sucrose. Subsequent work has shown that many alpha-amides of L-aspartic acid are sweet. Some results of stability studies and a taste panel evaluation of APM are reported.…
PMID: 994241
-
link
A novel route for aspartame production by combining enzymatic and chemical reactions for industrial use.
Here, we report a novel industrial aspartame production route, involving the enzymatic production of α-l-aspartyl-l-phenylalanine β-methylester from l-aspartic acid dimethylester and l-phenylalanine by α-amino acid ester acyl transferase. The route also involves the chemical transformation of α-l-aspartyl-l-phenylalanine β-methylester to α-l-aspartyl-l-phenylalanine methylester hydrochloride (aspartame hydrochloride) in an aqueous solution with methanol and HCl, followed by HCl removal to form aspartame.…
PMID: 33604621
"בזמן צריכה, מולקולת האספרטיים מתפרקת בגופנו לשתי חומצות אמינו ולמתנול."
מסקנת הבדיקה:
תהליך המטבוליזם של אספרטיים בגוף האדם מוביל לפירוקו לשלושה מרכיבים עיקריים: חומצה אספרטית, פנילאלנין ומתנול. (🟩)
chevron_right מקורות מדעיים: (2)
-
link
Aspartame and Its Potential Neurocognitive Effects in Humans.
The safety of aspartame, a widely consumed low-calorie artificial sweetener, for human consumption has recently been heavily debated. Published research has demonstrated that aspartame's breakdown products-aspartic acid, phenylalanine, and methanol-are associated with potential neurotoxicity and possible carcinogenic and metabolic effects in animal models. For this scoping review, existing literature on neurocognitive effects of aspartame was gathered and analyzed. A search of 3 literature databases (Embase, Ovid MEDLINE, and Web of Science) yielded 170 peer-reviewed, full-text articles. After consideration of exclusion criteria, 29 articles were selected for inclusion. After reviewing the findings, indications of aspartame's potential neurotoxic effects were demonstrated: multiple experimental studies revealed histopathological changes, such as elevated oxidative stress markers and neuronal loss, in various brain regions of animal models after exposure to aspartame. Additional studies showed memory and learning impairments in animal models, as well as behavioral dysfunction and mood disorders indicative of depression and anxiety in both animal and human models, illustrated by processes such as the downregulation of gamma aminobutyric acid signaling and upregulation of glutamate signaling in the amygdala. Notably, many of these studies investigated aspartame's impact not only at or above the Food and Drug Administration (FDA)-approved level, but below it as well. Two studies also indicated that populations with preexisting neurocognitive deficits, such as individuals with parkinsonism or preexisting metabolic conditions such as diabetes may have an increased vulnerability to neurocognitive harm when consuming aspartame. Interestingly, several agents were found to ameliorate aspartame's neurocognitive impacts, such as Pimpinella anisum oil and vitamin E. These negative neurocognitive effects and their potential mitigators demonstrate the necessity for further research into aspartame's possible impacts on health and safety at current FDA-approved levels, with additional consideration of certain populations at higher risk for neurocognitive injury.…
PMID: 40608001
-
link
Aspartame and ischemic stroke: unraveling the molecular link through network toxicology and molecular docking analysis.
Aspartame, a widely used artificial sweetener, remains controversial due to neurotoxic risks from its metabolites-phenylalanine, aspartic acid, and methanol. While epidemiological studies link artificial sweeteners to cerebrovascular disease, the molecular mechanisms connecting aspartame to ischemic stroke are unclear. This study integrates network toxicology and molecular docking to identify key targets and pathways. Potential aspartame targets were predicted using STITCH, SwissTargetPrediction, and SEA databases, while ischemic stroke-related genes were retrieved from GeneCards, OMIM, and TTD. Venn analysis identified 201 overlapping genes, with IL1B, MMP9, SRC, AGT, and TNF as core targets. GO/KEGG enrichment revealed their roles in the renin-angiotensin system, complement/coagulation cascades, and inflammatory pathways. Molecular docking demonstrated strong binding affinities between aspartame and these targets, suggesting direct modulation. Our integrated analysis suggests that aspartame may contribute to ischemic brain injury through multi-target interactions, potentially disrupting inflammatory responses and vascular homeostasis. This study provides preliminary systematic insights into the potential neurotoxicity mechanisms of aspartame, offering insights for food additive safety evaluation and stroke prevention. Further validation is required to clarify metabolite synergies and dose-response relationships.…
PMID: 40615604
"לפי ה-FDA, המינון הבטוח לשימוש באספרטיים הוא כ-50 מ"ג לכל קילוגרם משקל גוף ביום."
מסקנת הבדיקה:
ה-FDA קבע את הצריכה היומית המקובלת (ADI) של אספרטיים על 50 מ"ג לכל קילוגרם משקל גוף, בעוד ארגונים בינלאומיים אחרים כמו ה-WHO קבעו רף נמוך מעט יותר של 40 מ"ג. (🟩)
chevron_right מקורות מדעיים: (2)
-
link
Effects of aspartame on hsp70, bcl-2 and bax expression in immune organs of Wistar albino rats.
Aspartame, a "first generation sweetener", is widely used in a variety of foods, beverages, and medicine. The FDA has determined the acceptable daily intake (ADI) value of aspartame to be 50 mg/kg·day, while the JECFA (Joint FAO/WHO Expert Committee on Food Additives) has set this value at 40 mg/kg of body weight/day. Safety issues have been raised about aspartame due to its metabolites, specifically toxicity from methanol and/or its systemic metabolites formaldehyde and formic acid. The immune system is now recognized as a target organ for many xenobiotics, such as drugs and chemicals, which are able to trigger unwanted apoptosis or to alter the regulation of apoptosis. Our previous studies has shown that oral administration of aspartame [40 mg/(kg·day)] or its metabolites for 90 days increased oxidative stress in immune organs of Wistar albino rats. In this present study, we aimed to clarify whether aspartame consumption over a longer period (90-days) has any effect on the expression of hsp70, bcl-2 and bax at both mRNA transcript and protein expression levels in immune organs. We observed that oral administration of aspartame for 90 days did not cause any apparent DNA fragmentation in immune organs of aspartame treated animals; however, there was a significant increase in hsp70 expression, apart from significant alteration in bcl-2 and bax at both mRNA transcript and protein expression level in the immune organs of aspartame treated animals compared to controls. Hence, the results indicated that hsp70 levels increased in response to oxidative injury induced by aspartame metabolites; however, these metabolites did not induce apoptosis in the immune organs. Furthermore, detailed analyses are needed to elucidate the precise molecular mechanisms involved in these changes.…
PMID: 27845306
-
link
Acceptable daily intake vs actual intake: the aspartame example.
This article discusses the acceptable daily intake (ADI) and the postmarketing surveillance of consumption levels for a food additive, using the widely used food additive aspartame (APM, L-aspartyl-L-phenylalanine methyl ester) as an example. The safety implications of the ADI and consumption levels are also discussed. Aspartame has been assigned an ADI of 40 mg/kg/day by the World Health Organization and regulatory authorities in Europe and Canada, and of 50 mg/kg/day by the US Food and Drug Administration. A number of different methods have been used to measure consumption levels of food additives. Consumption estimations for aspartame from one such method, the food intake survey, have been done in the United States, Canada, Germany, and Finland. APM consumption in all age groups and selected subpopulations, even at the 90th percentile, is approximately 2-10 mg/kg/day and is thus well below the ADI.…
PMID: 1894884
"המיתוס על כך שאספרטיים גורם לסרטן מקורו בניסויים שנערכו בשנות ה-70 על חולדות, שדיווחו על עלייה בשכיחות גידולי מוח."
מסקנת הבדיקה:
החששות הראשוניים לגבי קשר בין אספרטיים לסרטן אכן נבעו ממחקרים מוקדמים בבעלי חיים בשנות ה-70, אשר העלו שאלות לגבי שכיחות גידולי מוח, אם כי מחקרים מאוחרים יותר לא אישרו ממצאים אלו באופן חד-משמעי. (🟩)
chevron_right מקורות מדעיים: (2)
-
link
NTP report on the toxicology studies of aspartame (CAS No. 22839-47-0) in genetically modified (FVB Tg.AC hemizygous) and B6.129-Cdkn2atm1Rdp (N2) deficient mice and carcinogenicity studies of aspartame in genetically modified [B6.129-Trp53tm1Brd (N5) haploinsufficient] mice (feed studies).
Aspartame is an artificial sweetener used throughout the world in food and beverages. Conventional 2-year rodent cancer studies of aspartame are considered negative, although a small number of neoplasms of the brain were observed in a rat study (Fed. Regist., 1981a,b). The NTP has explored the use of genetically altered mouse models as adjuncts to the 2-year rodent cancer assay. These models may prove to be more rapid, use fewer animals, and provide some mechanistic insights into neoplastic responses. As part of the evaluation of new mouse cancer screening models, aspartame was tested for potential toxicity and carcinogenicity in two relatively well-studied models, the Tg.AC hemizygous strain and the p53 haploinsufficient strain, and an uncharacterized model, the Cdkn2a deficient strain. Male and female Tg.AC hemizygous, p53 haploinsufficient, and Cdkn2a deficient mice were given feed containing aspartame (greater than 98% pure) for 9 months. Genetic toxicology studies were conducted in Salmonella typhimurium, rat bone marrow cells, and mouse peripheral blood erythrocytes. 9-MONTH STUDY IN Tg.AC HEMIZYGOUS MICE: Groups of 15 male and 15 female Tg.AC hemizygous mice were fed diets containing 0, 3,125, 6,250, 12,500, 25,000, or 50,000 ppm aspartame (equivalent to average daily doses of approximately 490, 980, 1,960, 3,960, or 7,660 mg aspartame/kg body weight to males and 550, 1,100, 2,260, 4,420, or 8,180 mg/kg to females) for 40 weeks. Exposure to aspartame had no effect on survival. The mean body weights of 50,000 ppm females were greater than those of the controls from week 15 until the end of the study. Feed consumption by the exposed groups was similar to that by the control groups throughout the study. There were no neoplasms or nonneoplastic lesions that were attributed to exposure to aspartame. 9-MONTH STUDY IN p53 HAPLOINSUFFICIENT MICE: Groups of 15 male and 15 female p53 haploinsufficient mice were fed diets containing 0, 3,125, 6,250, 12,500, 25,000, or 50,000 ppm aspartame (equivalent to average daily doses of approximately 490, 970, 1,860, 3,800, or 7,280 mg/kg to males and 630, 1,210, 2,490, 5,020, or 9,620 mg/kg to females) for 40 weeks. Exposure to aspartame had no effect on survival or mean body weights. Feed consumption by the exposed groups was similar to that by the control groups throughout the study. No neoplasms or nonneoplastic lesions were attributed to exposure to aspartame. 9-MONTH STUDY IN Cdkn2a DEFICIENT MICE: Groups of 15 male and 15 female Cdkn2a deficient mice were fed diets containing 0, 3,125, 6,250, 12,500, 25,000, or 50,000 ppm aspartame for 40 weeks (equivalent to average daily doses of approximately of approximately 490, 960, 1,900, 3,700, and 7,400 mg/kg to males and 610, 1,200, 2,390, 4,850, and 9,560 mg/kg to females). Survival of all exposed groups was similar to that of the control groups. Mean body weights of 3,125 and 6,250 ppm males were less than those of the controls after weeks 29 and 16, respectively. Mean body weights of female mice were similar to those of the controls throughout the study. The incidences of minimal to mild cytoplasmic vacuolization of periportal hepatocytes were significantly greater than controls in males exposed to 6,250, 25,000, or 50,000 ppm aspartame. Aspartame was tested for induction of gene mutations in Salmonella typhimurium. No mutagenicity was detected in strains TA98, TA100, or TA1535 with or without exogenous metabolic activation (S9). In addition, a single test in TA1537 with 30% rat liver S9 gave negative results. In TA97 with 30% rat liver S9, however, a reproducible small increase in mutant colonies was observed, and this response was judged to be equivocal. No mutagenicity was detected in TA97 without S9 or with hamster liver S9. An acute bone marrow micronucleus test was conducted with aspartame administered by gavage to male F344/N rats. No increase in micronucleated polychromatic erythrocytes was observed at any dose level. Peripheral blood micronucleus tests were conducted after 9 months exposure of Tg.AC hemizygous, p53 haploinsufficient, and Cdkn2a deficient mice to aspartame in dosed feed. Negative results were obtained in male and female Tg.AC hemizygous and Cdkn2a deficient mice. Negative results were also obtained with male p53 haploinsufficient mice. In female p53 haploinsufficient mice, the results of the micronucleus test were judged to be positive, based on a significant trend test and a small but statistically significant increased frequency of micronucleated erythrocytes in the 50,000 ppm group. Under the conditions of this 9-month feed study, there was no evidence of carcinogenic activity of aspartame in male or female p53 haploinsufficient mice exposed to 3,125, 6,250, 12,500, 25,000, or 50,000 ppm. Because this is a new model, there is uncertainty whether the study possessed sufficient sensitivity to detect a carcinogenic effect.…
PMID: 18685711
-
link
Histological analyses of the Ishii (1981) rat carcinogenicity study of aspartame and comparison with the Ramazzini Institute studies.
Researchers from the Ramazzini Institute have reported that lifespan dosing of rats with aspartame treatment is associated with an increased overall incidence of malignant tumors, including leukemias/lymphomas, transitional cell carcinomas of the renal pelvis/ureter, and malignant schwannomas of the peripheral nerves. Other carcinogenicity studies conducted on aspartame have shown no such carcinogenic potential in any organ system. Additional data to assess the carcinogenic potential of aspartame, especially in relation to the publications of the Ramazzini Institute, were obtained from a third-party histological evaluation of tissues from a carcinogenicity study previously conducted to assess the potential for aspartame to induce tumors of the brain. The results of this histological evaluation provide no evidence of a tumorigenic effect of aspartame in any organ group, including those organs/tissues reportedly affected in the Ramazzini Institute's studies. The only effects identified were an increased incidence of renal pelvic mineralization and renal pelvic hyperplasia secondary to the irritant properties of the mineralization process. The toxicological significance of these particular findings is widely considered minimal. There is no evidence that aspartame is carcinogenic in rats, at least to doses of 4 g/kg body weight/day administered over a 2-year period.…
PMID: 30572082
עירעור על דוח זה
ספקו ראיות חדשות או הצביעו על אי דיוקים
נעדכן אותך על תוצאות הבדיקה
הוסיפו קישורים למחקרים או מקורות רפואיים מוכרים
העירעור נשלח בהצלחה!
המנוע המדעי שלנו יבדוק את הראיות שהגשתם. נעדכן אתכם באימייל עם התוצאות.
ניתוח מבוסס בינה מלאכותית
דוח זה נוצר באופן אוטומטי על ידי מערכת בינה מלאכותית ועשוי להכיל שגיאות, אי-דיוקים או מידע חלקי. הניתוח אינו מהווה ייעוץ רפואי, אבחנה או המלצה לטיפול, והוא אינו תחליף לדעתו של איש מקצוע רפואי מוסמך. יש להתייעץ עם רופא או מומחה מוסמך לפני קבלת כל החלטה רפואית. המידע מוצג לצרכי מידע כללי בלבד.
מידע זה מופק על ידי בינה מלאכותית ואינו מהווה תחליף לייעוץ רפואי מקצועי.