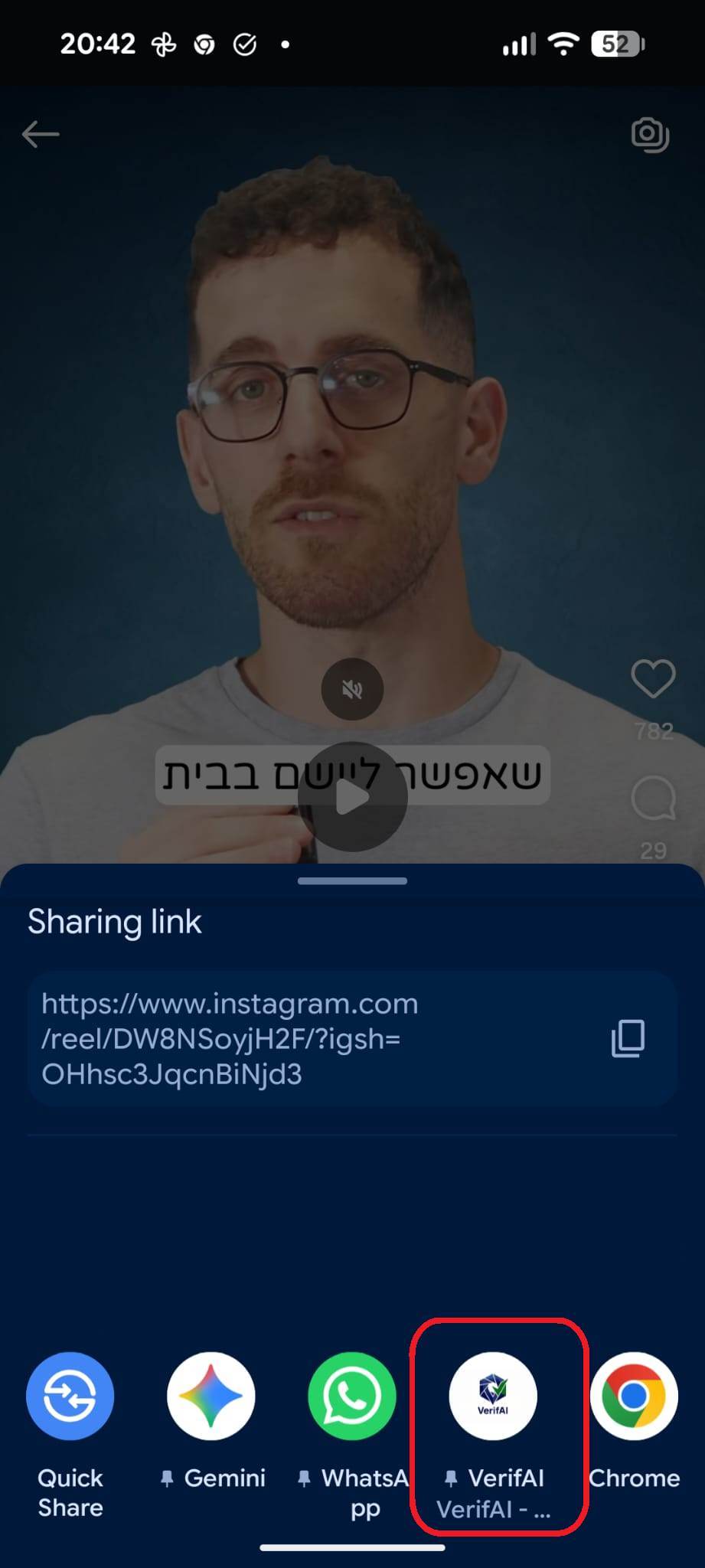
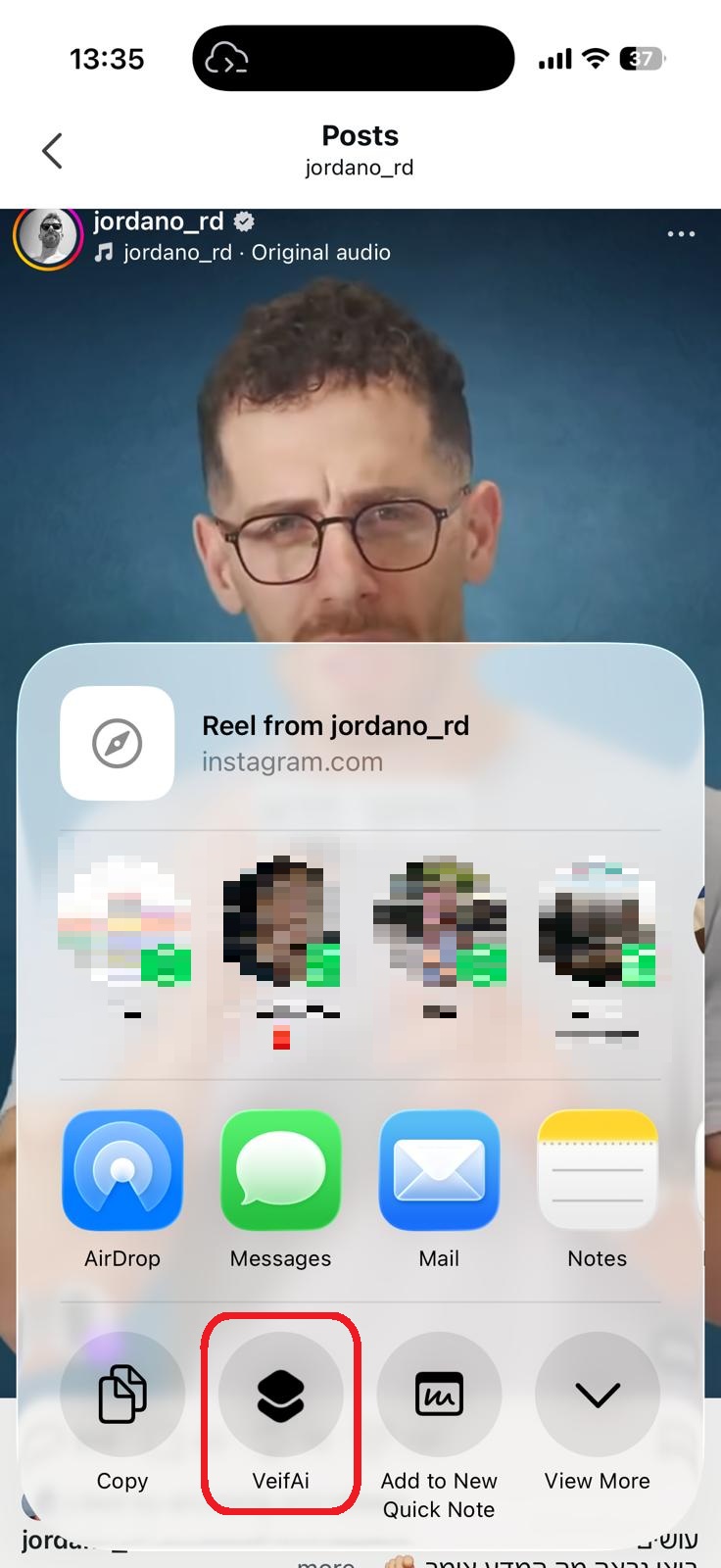
סריקה בתהליך...
הניתוח עשוי לארוך 30–90 שניות
הידעת?
הסרטון אינו מכיל תוכן רפואי
חושבים שחלה טעות?
אם לדעתכם הסרטון אכן עוסק בטענות רפואיות או בריאותיות, הוסיפו הסבר קצר ושלחו בקשה לבדיקה מחדש.
הבקשה התקבלה! נבדוק את הסרטון ונחזור אליכם.
אירעה שגיאה בשליחת הבקשה. נסו שוב.
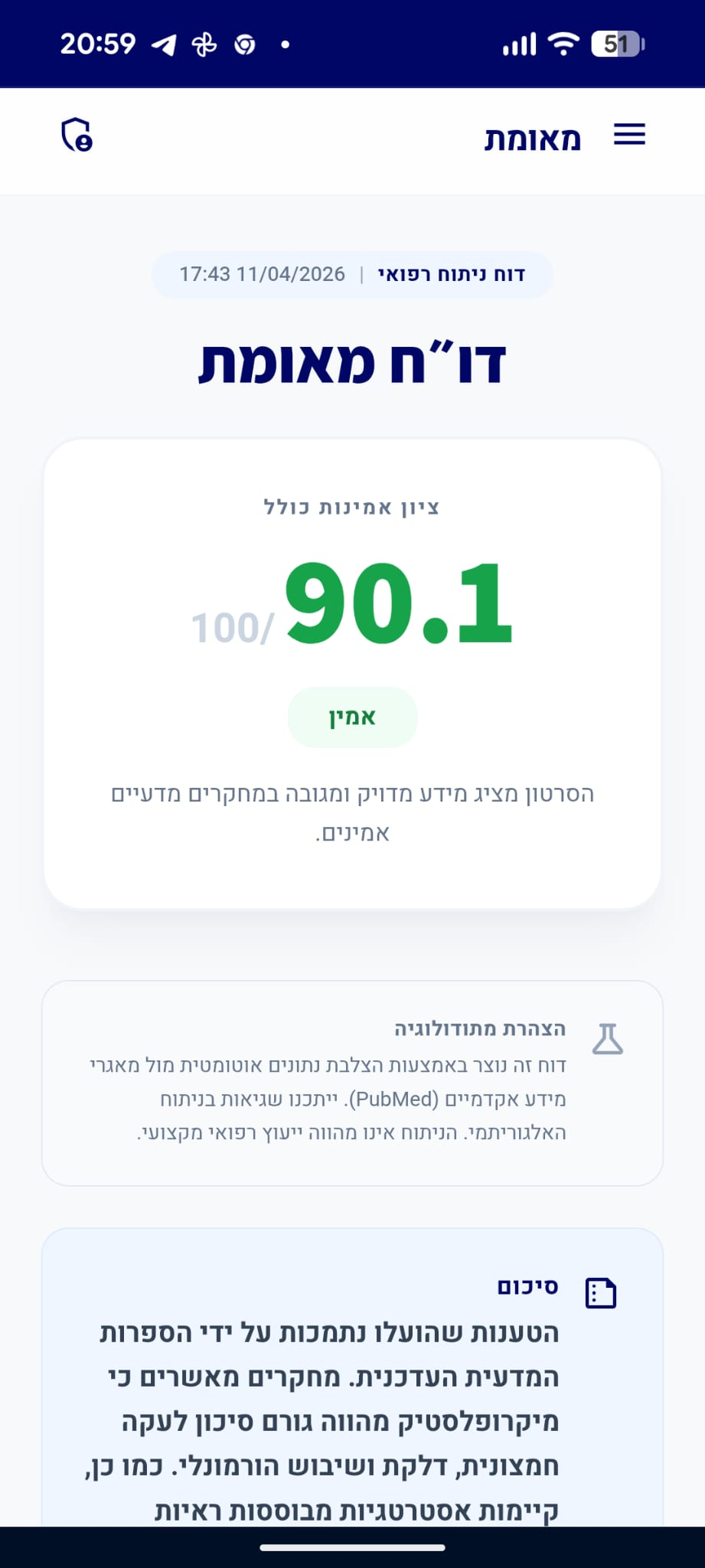
דו״ח מאומת
הסרטון מכיל שילוב של עובדות מוכחות וטענות המטעות את הציבור.
סיכום
הטענות המוצגות בוידאו תואמות ברובן את הקונצנזוס המדעי הנוכחי בנוגע לפלסטיות מוחית במהלך ההיריון. המדע מכיר בכך שמתרחשים שינויים מבניים (ירידה בנפח החומר האפור באזורים מסוימים) שאינם נזק מוחי אלא ארגון מחדש של רשתות עצביות, וחלקם נשמרים לאחר הלידה כהתאמה הורית. עם זאת, הנתון הספציפי של '4%' אינו מגובה במחקרים קליניים רחבים, והשימוש במונח 'התכווצות' עשוי להטעות מבחינה טרמינולוגית שכן הוא מרמז על פגיעה ולא על תהליך הסתגלותי.
analytics ניתוח טענות מבוסס ראיות
"המוח של האישה עובר תהליך של התכווצות (ירידה בנפח) במהלך ההיריון, ולוקח לו עד שישה חודשים לחזור לנפחו המקורי לאחר הלידה."
מסקנת הבדיקה:
הספרות המדעית מאשרת כי חלים שינויים בנפח החומר האפור במהלך ההיריון, אך הטענה לגבי 'התכווצות' (shrinkage) כתיאור מדויק של התהליך שנויה במחלוקת במינוח המדעי, שכן מדובר בארגון מחדש ולא באובדן נפח פתולוגי. בנוסף, אין עדות חד-משמעית לכך שלוקח בדיוק שישה חודשים לחזור לנפח המקורי, שכן מדובר בתהליך דינמי ומשתנה בין נשים. (🟨)
chevron_right מקורות מדעיים: (2)
-
link
Hormonal and structural transformations in the caregiver's brain: Examining themes in parental neurobiology.
When becoming a parent, caregivers undergo complex, and sometimes permanent, neurobiological alterations, and this area of neurobiology has been extensively studied for decades. Due to ethical concerns and experimental limitations, the first parental neurobiology experiments were exclusively performed using rodent animal model systems, such as mice, rats, and voles. More recent technological advancements, such as the functional MRI (fMRI) scan, have become widely adopted and led to great insight into the impact of parenting on human neurobiology. In this thematic literature review, we present key studies that provide insight into the relationship of pregnancy and parturition on maternal caregiving behavior and the relationship of postpartum on all parents. First, we examine the relationship of endocrine hormones such as estrogen, progesterone, oxytocin, and testosterone with the neurobiological development of a parent. Next, we describe the significant transformation of subcortical maternal circuit components that occur during pregnancy, and the changes in the volume of grey and white matter generated during the postpartum. These brain structure alterations contribute to the development of parental nurturing behaviors.…
PMID: 41523332
-
link
The Molecular and Cellular Basis of Physiological Changes in Pregnancy and Its Implications in Neurologic and Ophthalmic Pathologies.
Pregnancy orchestrates profound neurological, hormonal, and anatomical transformations in the maternal brain, preparing it for caregiving and infant bonding. Neuroimaging reveals structural changes such as gray matter reductions and white matter reorganization during pregnancy, followed by partial recovery postpartum. These adaptations are modulated by fluctuating levels of estradiol, progesterone, prolactin, and oxytocin, which coordinate neuroplasticity and behavioral readiness. At the molecular and cellular levels, pregnancy hormones drive synaptic remodeling, neurogenesis, and glial activity. Together, these changes support maternal motivation, attachment, and responsiveness, highlighting the maternal brain's dynamic plasticity across gestation and the postpartum period. Also, pregnancy induces profound physiological changes, particularly in vascular, hormonal, and neurologic systems, to support maternal and fetal health. While these adaptations are essential, they can predispose pregnant individuals to various neurologic and ophthalmic pathologies. This review explores how pregnancy-related changes-including hypercoagulability, pituitary enlargement, hormonal fluctuations, and immunological modulation-contribute to conditions such as stroke, idiopathic intracranial hypertension, preeclampsia-associated visual disturbances, and demyelinating disorders like neuromyelitis optica spectrum disorder and multiple sclerosis. Additionally, ocular manifestations of systemic diseases like diabetic retinopathy and thyroid orbitopathy are discussed. Understanding these complex interactions is critical for prompt recognition, accurate diagnosis, and appropriate management of vision-threatening and neurologically significant complications during pregnancy. Nevertheless, many aspects of physiological and pathological changes during and after pregnancy remain unknown and warrant further investigation.…
PMID: 40508034
"מחקרים מראים ירידה של כ-4% בחומר האפור באזורים מסוימים במוח במהלך ההיריון."
מסקנת הבדיקה:
אף שקיימות עדויות לירידה בנפח החומר האפור באזורים ספציפיים במוח במהלך ההיריון, לא נמצאו מחקרים המצביעים על נתון גורף של '4%' כשיעור קבוע או מייצג בספרות המדעית. השינויים הם אזוריים ומשתנים מאוד בין נשים. (⬜)
"הירידה בנפח המוח במהלך ההיריון אינה נובעת מנזק מוחי, אלא מתהליך של ארגון מחדש של רשתות עצביות."
מסקנת הבדיקה:
מחקרים נוירוביולוגיים עדכניים תומכים בכך שהשינויים המבניים במוח במהלך ההיריון אינם מעידים על נזק מוחי, אלא על תהליכי פלסטיות מוחית והתאמה של רשתות עצביות (כגון ה-Default Mode Network) לצורכי ההורות והטיפול בתינוק. (🟩)
chevron_right מקורות מדעיים: (3)
-
link
Hormonal and structural transformations in the caregiver's brain: Examining themes in parental neurobiology.
When becoming a parent, caregivers undergo complex, and sometimes permanent, neurobiological alterations, and this area of neurobiology has been extensively studied for decades. Due to ethical concerns and experimental limitations, the first parental neurobiology experiments were exclusively performed using rodent animal model systems, such as mice, rats, and voles. More recent technological advancements, such as the functional MRI (fMRI) scan, have become widely adopted and led to great insight into the impact of parenting on human neurobiology. In this thematic literature review, we present key studies that provide insight into the relationship of pregnancy and parturition on maternal caregiving behavior and the relationship of postpartum on all parents. First, we examine the relationship of endocrine hormones such as estrogen, progesterone, oxytocin, and testosterone with the neurobiological development of a parent. Next, we describe the significant transformation of subcortical maternal circuit components that occur during pregnancy, and the changes in the volume of grey and white matter generated during the postpartum. These brain structure alterations contribute to the development of parental nurturing behaviors.…
PMID: 41523332
-
link
The Molecular and Cellular Basis of Physiological Changes in Pregnancy and Its Implications in Neurologic and Ophthalmic Pathologies.
Pregnancy orchestrates profound neurological, hormonal, and anatomical transformations in the maternal brain, preparing it for caregiving and infant bonding. Neuroimaging reveals structural changes such as gray matter reductions and white matter reorganization during pregnancy, followed by partial recovery postpartum. These adaptations are modulated by fluctuating levels of estradiol, progesterone, prolactin, and oxytocin, which coordinate neuroplasticity and behavioral readiness. At the molecular and cellular levels, pregnancy hormones drive synaptic remodeling, neurogenesis, and glial activity. Together, these changes support maternal motivation, attachment, and responsiveness, highlighting the maternal brain's dynamic plasticity across gestation and the postpartum period. Also, pregnancy induces profound physiological changes, particularly in vascular, hormonal, and neurologic systems, to support maternal and fetal health. While these adaptations are essential, they can predispose pregnant individuals to various neurologic and ophthalmic pathologies. This review explores how pregnancy-related changes-including hypercoagulability, pituitary enlargement, hormonal fluctuations, and immunological modulation-contribute to conditions such as stroke, idiopathic intracranial hypertension, preeclampsia-associated visual disturbances, and demyelinating disorders like neuromyelitis optica spectrum disorder and multiple sclerosis. Additionally, ocular manifestations of systemic diseases like diabetic retinopathy and thyroid orbitopathy are discussed. Understanding these complex interactions is critical for prompt recognition, accurate diagnosis, and appropriate management of vision-threatening and neurologically significant complications during pregnancy. Nevertheless, many aspects of physiological and pathological changes during and after pregnancy remain unknown and warrant further investigation.…
PMID: 40508034
-
link
Temporal dissociation between local and global functional adaptations of the maternal brain to childbirth: a longitudinal assessment.
The maternal brain undergoes significant reorganization during birth and the postpartum period. However, the temporal dynamics of these changes remain unclear. Using resting-state functional magnetic resonance imaging, we report on local and global brain function alterations in 75 mothers in their first postpartum week, compared to 23 nulliparous women. In a subsample followed longitudinally for the next six months, we observed a temporal and spatial dissociation between changes observed at baseline (cluster mass permutation: pFWE < 0.05). Local activity and connectivity changes in widespread neocortical regions persisted throughout the studied time period (ANCOVAs vs. controls: pFDR < 0.05), with preliminary evidence linking these alterations to behavioral and psychological adaptations (interaction effect with postpartum time: uncorrected p < 0.05). In contrast, the initially reduced whole-brain connectivity of putamen-centered subcortical areas returned to control levels within six to nine weeks postpartum (linear and quadratic mixed linear models: pFDR < 0.05). The whole-brain spatial colocalization with hormone receptor distributions (Spearman correlations: pFDR < 0.05) and preliminary blood hormone associations (interaction effect with postpartum time: uncorrected p < 0.05) suggested that the postpartum restoration of progesterone levels may underlie this rapid normalization. These observations enhance our understanding of healthy maternal brain function, contributing to the identification of potential markers for pathological postpartum adaptation processes, which in turn could underlie postpartum psychiatric disorders.…
PMID: 38769432
"חלק מהשינויים בחומר האפור במוח נשארים גם לאחר הלידה, מה שמרמז על התאמות מוחיות קבועות לדרישות ההורות."
מסקנת הבדיקה:
הספרות המדעית מצביעה על כך שחלק מהשינויים המבניים והתפקודיים במוח נשמרים גם לאחר הלידה, מה שתומך בהשערה שמדובר בהתאמות נוירוביולוגיות ארוכות טווח המכינות את המוח לתפקידי ההורות. (🟩)
chevron_right מקורות מדעיים: (3)
-
link
Hormonal and structural transformations in the caregiver's brain: Examining themes in parental neurobiology.
When becoming a parent, caregivers undergo complex, and sometimes permanent, neurobiological alterations, and this area of neurobiology has been extensively studied for decades. Due to ethical concerns and experimental limitations, the first parental neurobiology experiments were exclusively performed using rodent animal model systems, such as mice, rats, and voles. More recent technological advancements, such as the functional MRI (fMRI) scan, have become widely adopted and led to great insight into the impact of parenting on human neurobiology. In this thematic literature review, we present key studies that provide insight into the relationship of pregnancy and parturition on maternal caregiving behavior and the relationship of postpartum on all parents. First, we examine the relationship of endocrine hormones such as estrogen, progesterone, oxytocin, and testosterone with the neurobiological development of a parent. Next, we describe the significant transformation of subcortical maternal circuit components that occur during pregnancy, and the changes in the volume of grey and white matter generated during the postpartum. These brain structure alterations contribute to the development of parental nurturing behaviors.…
PMID: 41523332
-
link
Temporal dissociation between local and global functional adaptations of the maternal brain to childbirth: a longitudinal assessment.
The maternal brain undergoes significant reorganization during birth and the postpartum period. However, the temporal dynamics of these changes remain unclear. Using resting-state functional magnetic resonance imaging, we report on local and global brain function alterations in 75 mothers in their first postpartum week, compared to 23 nulliparous women. In a subsample followed longitudinally for the next six months, we observed a temporal and spatial dissociation between changes observed at baseline (cluster mass permutation: pFWE < 0.05). Local activity and connectivity changes in widespread neocortical regions persisted throughout the studied time period (ANCOVAs vs. controls: pFDR < 0.05), with preliminary evidence linking these alterations to behavioral and psychological adaptations (interaction effect with postpartum time: uncorrected p < 0.05). In contrast, the initially reduced whole-brain connectivity of putamen-centered subcortical areas returned to control levels within six to nine weeks postpartum (linear and quadratic mixed linear models: pFDR < 0.05). The whole-brain spatial colocalization with hormone receptor distributions (Spearman correlations: pFDR < 0.05) and preliminary blood hormone associations (interaction effect with postpartum time: uncorrected p < 0.05) suggested that the postpartum restoration of progesterone levels may underlie this rapid normalization. These observations enhance our understanding of healthy maternal brain function, contributing to the identification of potential markers for pathological postpartum adaptation processes, which in turn could underlie postpartum psychiatric disorders.…
PMID: 38769432
-
link
The Molecular and Cellular Basis of Physiological Changes in Pregnancy and Its Implications in Neurologic and Ophthalmic Pathologies.
Pregnancy orchestrates profound neurological, hormonal, and anatomical transformations in the maternal brain, preparing it for caregiving and infant bonding. Neuroimaging reveals structural changes such as gray matter reductions and white matter reorganization during pregnancy, followed by partial recovery postpartum. These adaptations are modulated by fluctuating levels of estradiol, progesterone, prolactin, and oxytocin, which coordinate neuroplasticity and behavioral readiness. At the molecular and cellular levels, pregnancy hormones drive synaptic remodeling, neurogenesis, and glial activity. Together, these changes support maternal motivation, attachment, and responsiveness, highlighting the maternal brain's dynamic plasticity across gestation and the postpartum period. Also, pregnancy induces profound physiological changes, particularly in vascular, hormonal, and neurologic systems, to support maternal and fetal health. While these adaptations are essential, they can predispose pregnant individuals to various neurologic and ophthalmic pathologies. This review explores how pregnancy-related changes-including hypercoagulability, pituitary enlargement, hormonal fluctuations, and immunological modulation-contribute to conditions such as stroke, idiopathic intracranial hypertension, preeclampsia-associated visual disturbances, and demyelinating disorders like neuromyelitis optica spectrum disorder and multiple sclerosis. Additionally, ocular manifestations of systemic diseases like diabetic retinopathy and thyroid orbitopathy are discussed. Understanding these complex interactions is critical for prompt recognition, accurate diagnosis, and appropriate management of vision-threatening and neurologically significant complications during pregnancy. Nevertheless, many aspects of physiological and pathological changes during and after pregnancy remain unknown and warrant further investigation.…
PMID: 40508034

ישראל בריאות
דירוג זה מבוסס על 1 דוחות אימות קודמים.
האם הדוח הזה היה מועיל לך?
מה היה פחות טוב? (רשות)
תודה על הפידבק!
עירעור על דוח זה
ספקו ראיות חדשות או הצביעו על אי דיוקים
נעדכן אותך על תוצאות הבדיקה
הוסיפו קישורים למחקרים או מקורות רפואיים מוכרים
העירעור נשלח בהצלחה!
המנוע המדעי שלנו יבדוק את הראיות שהגשתם. נעדכן אתכם באימייל עם התוצאות.
ניתוח מבוסס בינה מלאכותית
דוח זה נוצר באופן אוטומטי על ידי מערכת בינה מלאכותית ועשוי להכיל שגיאות, אי-דיוקים או מידע חלקי. הניתוח אינו מהווה ייעוץ רפואי, אבחנה או המלצה לטיפול, והוא אינו תחליף לדעתו של איש מקצוע רפואי מוסמך. יש להתייעץ עם רופא או מומחה מוסמך לפני קבלת כל החלטה רפואית. המידע מוצג לצרכי מידע כללי בלבד.
מידע זה מופק על ידי בינה מלאכותית ואינו מהווה תחליף לייעוץ רפואי מקצועי.